|
The new results show that, as expected, flerovium is inert but capable of forming stronger chemical bonds than noble gases, if conditions are suitable. The two experiments were unable to clearly establish the character. While the three atoms observed in the first experiment were used to infer noble gas-like behavior, the data obtained at GSI indicated metallic character based on two atoms. Two previously conducted chemistry experiments, one of them at GSI in Darmstadt in 2009, led to contradictory interpretations. 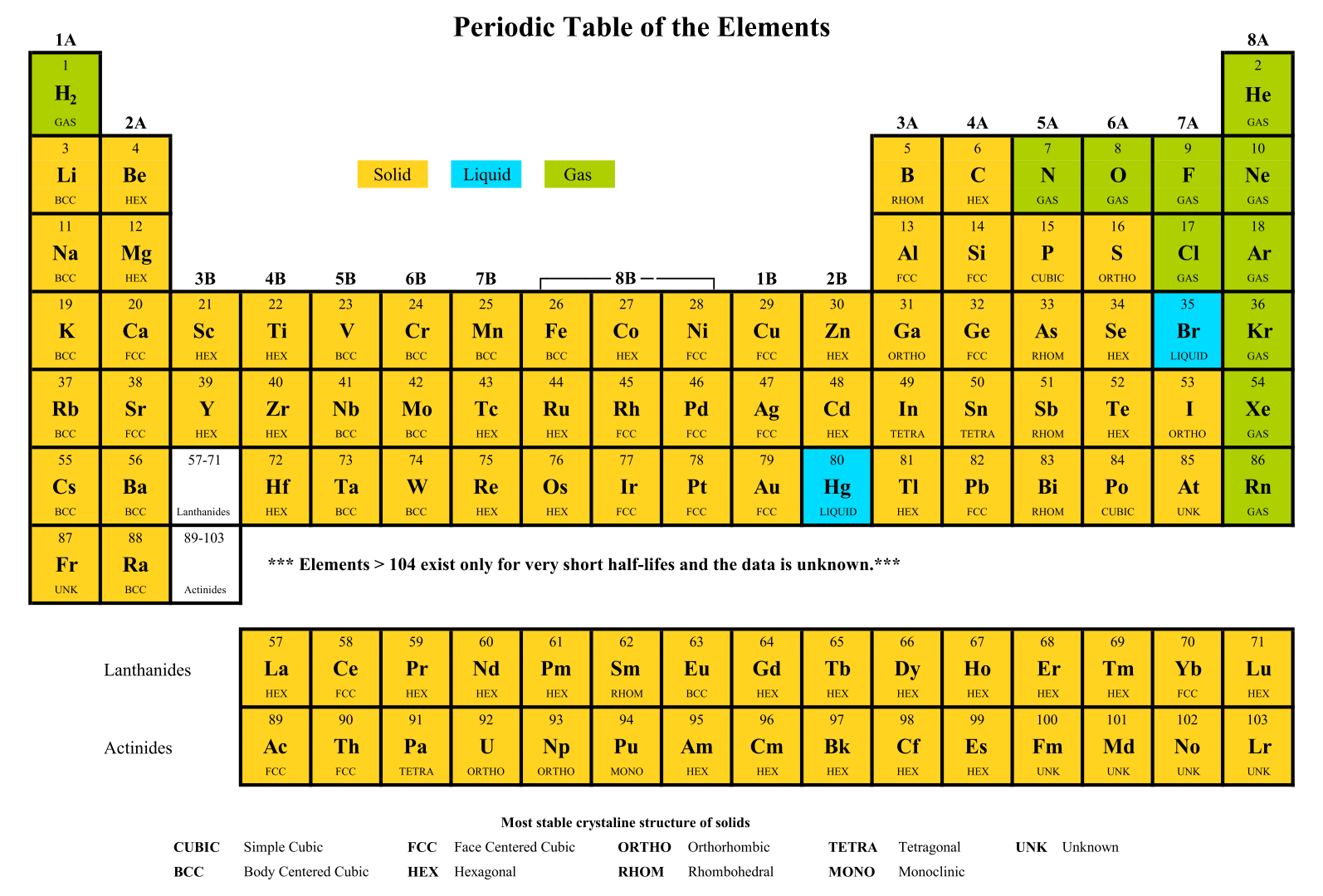
However, early predictions had postulated that relativistic effects of the high charge in the nucleus of the superheavy element on its valence electrons would lead to noble gas-like behavior, while more recent ones had rather suggested a weakly metallic behavior. In the periodic table, flerovium is placed below the heavy metal lead. 
Under the leadership of groups from Darmstadt and Mainz, the two longest-lived flerovium isotopes currently known, flerovium-288 and flerovium-289, were produced using the accelerator facilities at GSI/FAIR and were chemically investigated at the TASCA experimental setup. With the results, published in the journal Frontiers in Chemistry, GSI contributes to the study of the chemistry of superheavy elements and opens new perspectives for the international facility FAIR (Facility for Antiproton and Ion Research), which is currently under construction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
